11, the Food and Drug Administration granted the vaccine, known as Comirnaty, the first emergency use authorization ever given by the United States to a coronavirus vaccine. It was the first time anyone had found such evidence. 9, 2020, New York-based Pfizer and the German company BioNTech made history by announcing that their coronavirus vaccine had an efficacy rate of over 90 percent, far surpassing expectations. Storage: Freezer storage only at –13☏ to 5☏ (–25☌ to –15☌) Vaccine name: Comirnaty (also known as tozinameran or BNT162b2) Trials may also be abandoned if they indicate a vaccine isn’t effective against Covid-19.ĪPPROVED IN U.S., ELSEWHERE EMERGENCY USE IN MANY COUNTRIES After an investigation, the trial may resume or be abandoned. 
PAUSED or ABANDONED: If investigators observe worrying symptoms in volunteers, they can pause the trial. Some vaccines are now in Phase 1/2 trials, for example, which this tracker would count as both Phase 1 and Phase 2. Experts have warned of serious risks from jumping ahead of these results.ĪPPROVAL: Regulators review the complete trial results and plans for a vaccine’s manufacturing, and decide whether to give it full approval.ĬOMBINED PHASES: One way to accelerate vaccine development is to combine phases. In addition, some countries such as China and Russia began administering vaccines before detailed Phase 3 trial data was made public. Phase 3 trials are also large enough to reveal evidence of relatively rare side effects.ĮARLY OR LIMITED APPROVAL: Many countries have procedures for providing emergency authorizations for vaccines, based on preliminary evidence that they are safe and effective. These trials can determine if the vaccine protects against the coronavirus, measuring what’s known as the efficacy rate. PHASE 3 EFFICACY TRIALS: Scientists give the vaccine to thousands of people and wait to see how many become infected, compared with volunteers who received a placebo. These trials further test the vaccine’s safety. PHASE 2 EXPANDED TRIALS: Scientists give the vaccine to hundreds of people split into groups, such as children and the elderly, to see if the vaccine acts differently in them. PHASE 1 SAFETY TRIALS: Scientists give the vaccine to a small number of people to test safety and dosage, as well as to confirm that it stimulates the immune system. PRECLINICAL TESTING: Scientists test a new vaccine on cells and then give it to animals such as mice or monkeys to see if it produces an immune response. The development cycle of a vaccine, from lab to clinic. For an explanation of leading vaccines, see How Nine Covid-19 Vaccines Work. For treatments for Covid-19, see our Coronavirus Drug and Treatment Tracker. 
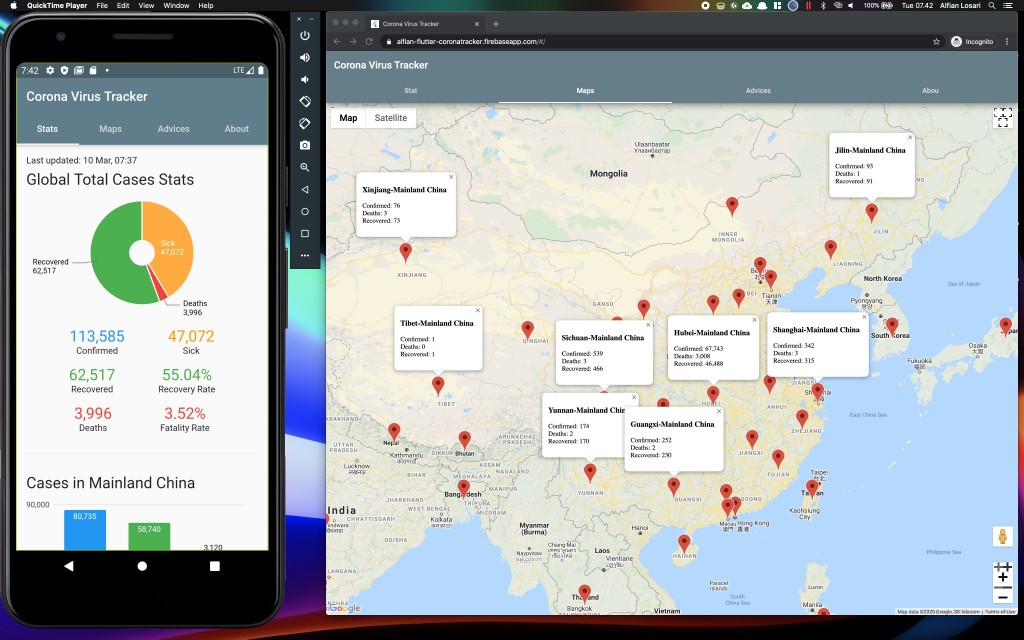
For an explanation of virus variants and mutations, see our Coronavirus Variant Tracker. The incubation period of severe acute respiratory syndrome coronavirus 2( SARS-CoV-2) infection is assumed to be 14 days succeeding exposure, mostly around four to five days.Below is a list of all vaccines that have reached trials in humans. Coronavirus disease 2019 (COVID-19) is spread through large droplets produced during coughing and sneezing by symptomatic patients, as well as asymptomatic individuals before starting of their symptoms. The novel coronavirus has rapidly become widespread, resulting in an epidemic throughout China, followed by a pandemia, an increasing number of cases in various countries throughout the world. At the end of December 2019, a novel coronavirus was recognized as the reason for a group of pneumonia cases of unidentified etiology in Wuhan, a city in the Hubei Province of China. The coronavirus family has significant human and animal pathogens.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
